The Pharmacogenomics Medical Devices Industry
The Pharmacogenomics Medical Devices Industry is a rapidly evolving field that integrates genetic information into clinical practice to enhance patient care.
The Pharmacogenomics Medical Devices Industry is composed of various specialised organisations that may deliver one or more steps in the clinical pathway or provide a complete end-to-end service. The organisations that form the foundation of the pharmacogenomics industry include commercial laboratories, medical device manufacturers (laboratory, scientific, software), innovators and service providers. These organisations operate across multiple industry and professional bodies, including:
- Association of British Pharmaceutical Industry (ABPI)
- Association of British Health Tech Industries (ABHI)
- British In Vitro Diagnostics Association (BIVDA)
- Institute of Biomedical Scientists (IBMS)
The UK-IPN has been formed to bring together these diverse stakeholders and promote the implementation of pharmacogenomic services in the UK.
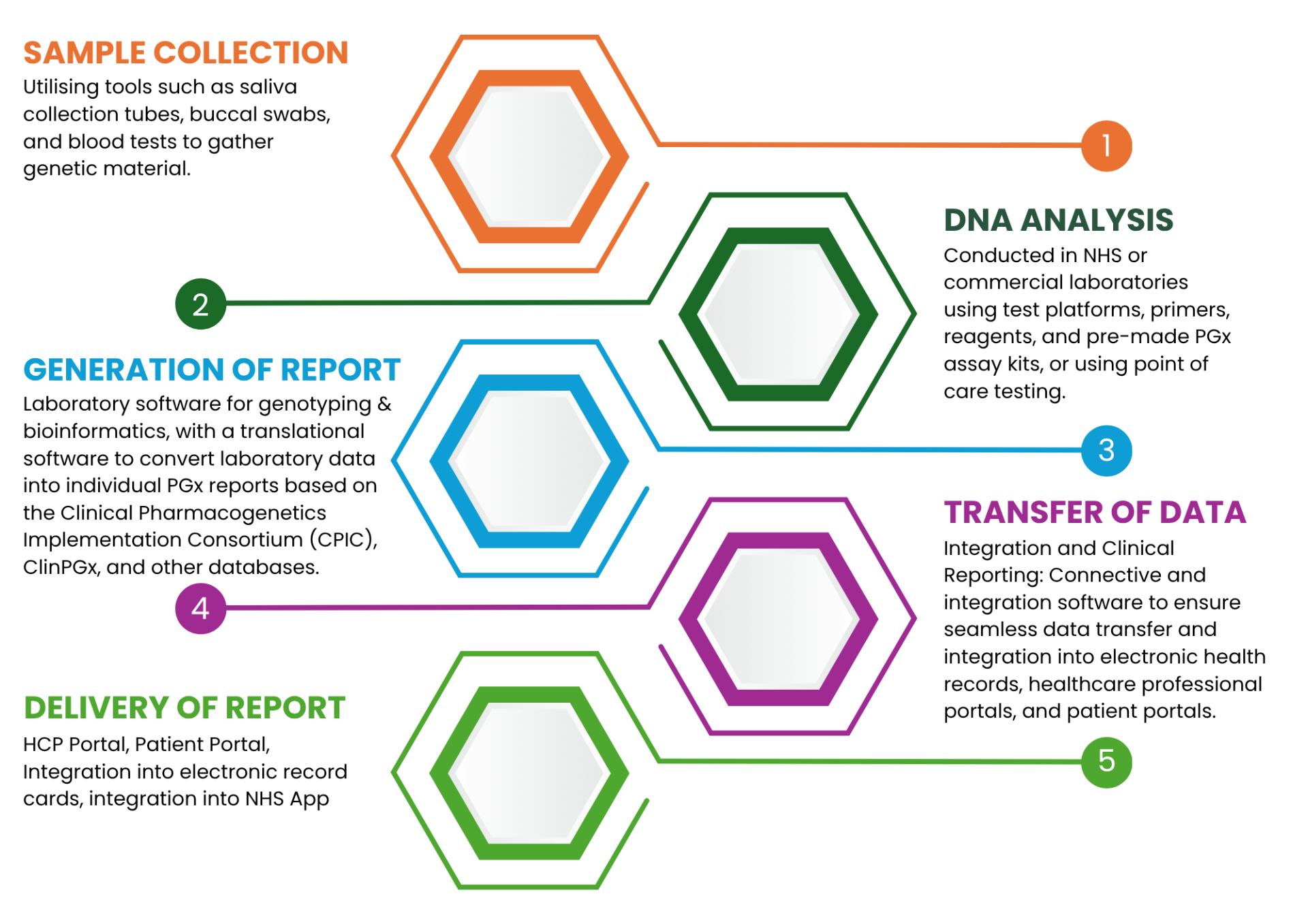
What medical devices are required to deliver pharmacogenomic (PGx) testing?
Our Commitment
At UK-IPN, we are dedicated to demonstrating leadership and professional standards in the PGx industry. We strive to implement PGx testing as a standard of care for all patients, working in partnership with the NHS, government, and regulatory organisations to identify and overcome barriers to the adoption of PGx testing.

